Our extensive patient resources include tools, guides and support videos – everything youneed to help you get the healing results you want with EXOGEN.
All revisions of the Instructions for Use can be made available upon request. Contact [email protected] or call 0800 05 16 384 to request an electronic copy.
EXOGEN Performance Program
Detailed information on the EXOGEN Performance Program, guidelines and requirements.
EXOGEN CONNECTS App
EXOGEN CONNECTS is a free smartphone app that sends automated treatment reminders and provides helpful healing information.
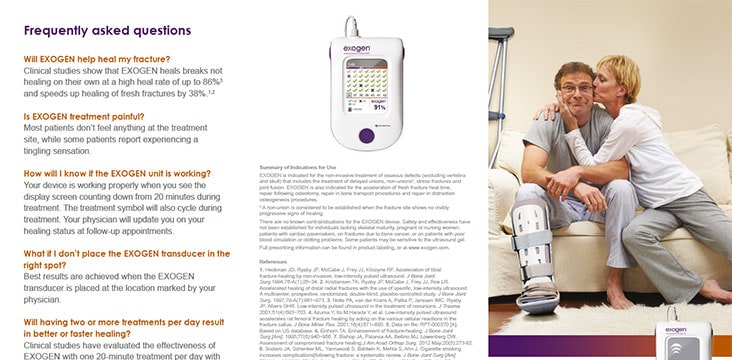
EXOGEN Patient Brochure
Introduction to the EXOGEN Ultrasound Bone Healing System, with answers to frequently asked questions.
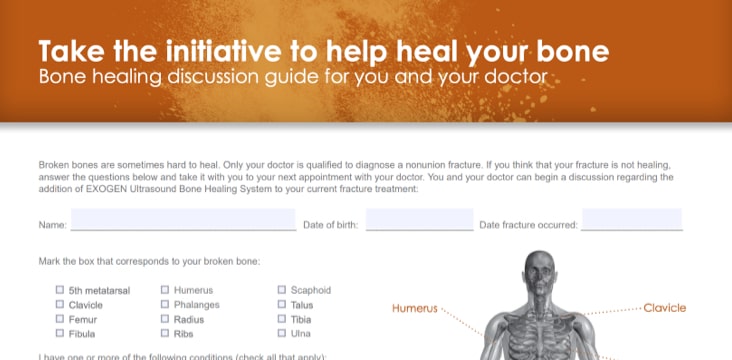
EXOGEN Bone Healing Discussion Guide
Assessment tool to determine if EXOGEN is right for you.
How to Use EXOGEN
View the How to Use EXOGEN video for a step-by-step guide on using EXOGEN.
Summary of Indications for Use
The EXOGEN Ultrasound Bone Healing System is indicated for the non-invasive treatment of established nonunions* excluding skull and vertebra. The EXOGEN device has also been reported as effective as an adjunctive non-invasive treatment of established nonunions in patients:
- With internal or external fracture fixation hardware present. EXOGEN cannot penetrate metal and therefore should not be applied directly over hardware.
- Undergoing treatment for infection at the fracture site. EXOGEN is not intended to treat the infection.
- Believed to have diminished bone quality. EXOGEN is not intended to treat diminished bone quality.
In addition, EXOGEN is indicated for accelerating the time to a healed fracture for fresh, closed, posteriorly displaced distal radius fractures and fresh, closed or Grade I open tibial diaphysis fractures in skeletally mature individuals when these fractures are orthopedically managed by closed reduction and cast immobilization.
There are no known contraindications for the EXOGEN device. Safety and effectiveness have not been established for individuals lacking skeletal maturity, pregnant or nursing women, patients with cardiac pacemakers, on fractures due to bone cancer, or on patients with poor blood circulation or clotting problems. Some patients may be sensitive to the ultrasound gel.
*A nonunion is considered to be established when the fracture site shows no visibly progressive signs of healing.
Full prescribing information can be found in product labelling, at EXOGEN.com, or by calling Customer Service at 1-800-836-4080.